Speaking at the ANS Annual Conference executive session on “How Nuclear Technologies will Shape the Future Energy Economy” were (from left) Craig Piercy, Stephen Carmel, Rian Bahran, Ross Radel, Greg Schulze, Harsh Desai, and Kirt Marlow.
The applications of nuclear energy extend beyond providing power to the electrical grid. Advanced nuclear technologies may soon have new applications in oil and gas facilities, in hospitals and clinics, on the open seas, and on the moon.
A June 1 executive session, “How Nuclear Technologies will Shape the Future Energy Economy,” at the American Nuclear Society’s Annual Conference allowed experts have an open discussion on the future of nuclear advancements in multiple sectors.
The Shine Chrysalis isotope production facility under construction in 2024. (Image: Shine)
Fusion technology company Shine has been issued a conditional commitment for a loan of up to $263 million by the Department of Energy’s Office of Energy Dominance Financing (EDF) to support the construction of the company’s medical isotope production facility in Janesville, Wis.
Orano USA CEO Jean-Luc Palayer (middle) shakes hands with Zeno Power’s cofounder and CEO Tyler Bernstein (left) and Chief Commercialization Officer Harsh Desai. (Photo: Orano USA)
Zeno Power, a developer of nuclear batteries, is to receive americium-241 recovered from Orano’s La Hague nuclear fuel recycling site in Normandy, France, under a strategic agreement announced by the companies on September 24.
Argonne’s Peter Tkac (left) and David Bettinardi analyze results from lab experiments designed to isolate desirable products from spent nuclear fuel. (Photo: ANL)
The Department of Energy’s Argonne National Laboratory will collaborate with Wisconsin-based fusion technology company Shine to design new chemical processes for separating valuable materials from used nuclear fuel.
Orano CEO Nicolas Maes (left) and SHINE Technologies founder and CEO Greg Piefer shake hands after agreeing to cooperate on a pilot used fuel recycling facility. (Photo: Orano)
Orano and SHINE Technologies have agreed to cooperate in the development of a pilot plant capable of recycling used nuclear fuel from light water reactors on a commercial scale. In announcing the signing of a memorandum of understanding on Thursday, the companies said the selection of a site for the pilot U.S. facility is expected by the end of this year.
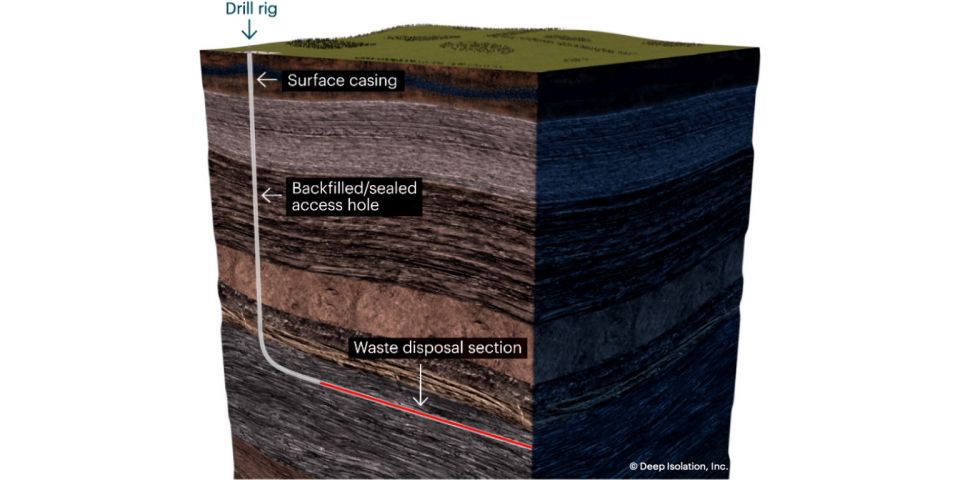
Schematic of a deep horizontal borehole repository for nuclear waste. (Image: Deep Isolation)
Waste disposal start-up Deep Isolation and fusion tech company SHINE Technologies have announced the completion of a collaborative study assessing the costs of disposing of radioactive byproducts from a pilot spent nuclear fuel recycling facility.
A vial of Ac-225 produced by Niowave stands next to its lead shipping pig. (Photo: Niowave)
According to the Council on Radionuclides and Radiopharmaceuticals, more than 82,000 nuclear imaging procedures using nuclear medicine are performed throughout the world every day. To administer these vital medical procedures, radiopharmaceutical companies and hospitals rely on a handful of producers of medical radioisotopes.
A still from a video on Westinghouse Electric Company’s eVinci microreactor, one of seven advanced reactor technologies that received support in GAIN’s latest round of nuclear energy vouchers. (Image: Westinghouse)
The Gateway for Accelerated Innovation in Nuclear (GAIN) announced December 19 that seven firms will get vouchers to access the nuclear research facilities and expertise of the national laboratory complex in the first round of fiscal year awards. Each company is paired with one or more national laboratories to work on concepts from advanced reactor fueling to fuel recycling to climate forecasting.
A spent nuclear fuel transportation container. (Photo: DOE)
Fusion systems company SHINE Technologies has notified the Nuclear Regulatory Commission that it intends to submit a license application to build and operate a pilot used nuclear fuel recycling facility.
SHINE Technologies founder and CEO Greg Piefer shows the hot cell banks that will be used to produce Lu-177. (Image: SHINE)
Fusion tech company SHINE Technologies announced that it is opening the largest facility in North America dedicated to the production of non-carrier-added lutetium-177, a medical isotope used in targeted cancer therapies.
SHINE’s Mo-99 production building under construction in October 2022. (Photo: SHINE)
Fusion development company SHINE Technologies announced that it will begin offering radiation effects testing in a dedicated facility on the company’s Janesville, Wis., campus later this year. SHINE will use high-energy fusion neutrons to test mission-critical components that are susceptible to radiation-harsh environments on behalf of its aerospace and defense customers.
The electron accelerator that will be used for Mo-99 production at NorthStar’s newly completed facility in Wisconsin. (Photo: NNSA)
NorthStar Medical Radioisotopes has completed construction and all equipment installation at its new facility in Beloit, Wis., to produce the medical radioisotope molybdenum-99 without the use of high-enriched uranium, the Department of Energy’s National Nuclear Security Administration announced last week.
A rendering of the SHINE medical isotope production facility planned for construction in Veendam, the Netherlands. (Image: SHINE)
SHINE Europe, a subsidiary of Wisconsin-based SHINE Technologies, will work with the Netherlands’ University Medical Center Groningen (UMCG) and Delft University of Technology (TU Delft) to produce a variety of terbium isotopes for use in nuclear medicine under a grant proposal approved by the Dutch government on October 17.










.png)

