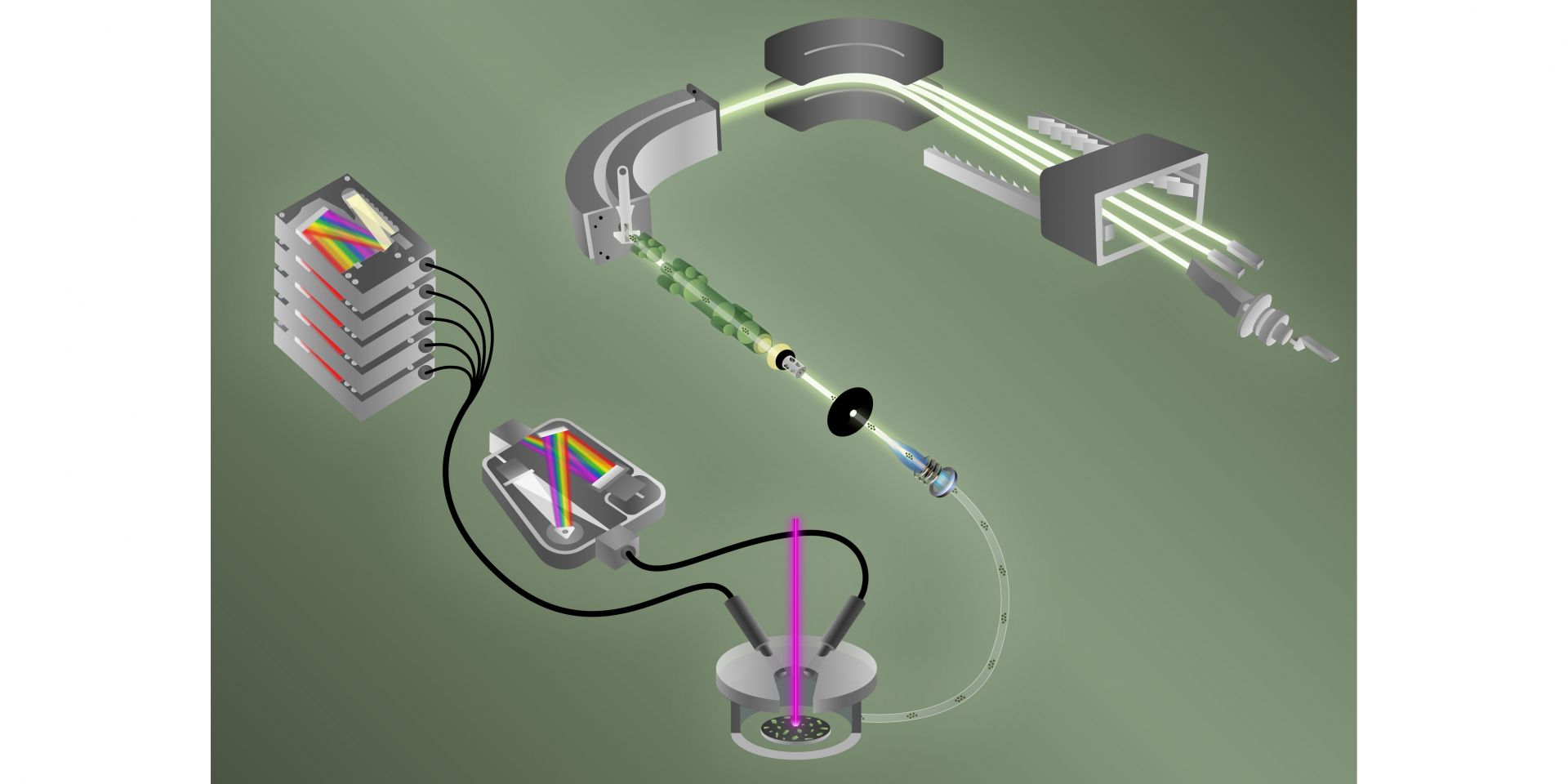
ORNL’s tandem technologies detect fluorine and isotopes of uranium at the same time to discern the fingerprint of a nuclear material made for fuel or weaponry. (Image: Benjamin Manard and Jacquelyn DeMink/ORNL)
By combining two techniques, analytical chemists at Oak Ridge National Laboratory have for the first time simultaneously detected fluorine and different uranium isotopes in a single particle. Quickly detecting both elements together may help International Atomic Energy Agency inspectors determine if and when undisclosed enrichment has taken place. The findings, published in the Journal of the American Chemical Society, “push the limit” of how fast single particles can be characterized in terms of their chemical, elemental, and isotopic compositions, according to a September 26 news release from ORNL.
Scientist Jacklyn Gates at the Berkeley Gas-filled Separator used to separate atoms of element 116, livermorium. (Photo: Marilyn Sargent/Berkeley Lab)
A plutonium target bombarded with a beam of titanium-50 in Lawrence Berkeley National Laboratory’s 88-Inch Cyclotron for 22 days has yielded two atoms of the superheavy element 116, in a proof of concept that gives Berkeley Lab researchers a path to pursue the heaviest element yet—element 120. The result was announced July 23 at the Nuclear Structure 2024 conference; a paper has been submitted to the journal Physical Review Letters and published on arXiv.
Jamie Weaver with the neutron depth profiling instrument. (Photo: T. Barvitskie/NIST)
The newest generation of lithium-ion batteries now being developed uses thin-film, solid-state technology and could soon safely power cell phones, electric vehicles, laptops, and other devices. However, like all batteries, solid-state lithium-ion batteries have a drawback: Impedance—electrical resistance—can build up as batteries are discharged and recharged, limiting the flow of electric current.
Artist’s view of heavy water eliciting sweet taste in humans. Graphic design: Tomáš Bello/IOCB Prague
Is isotope science all sweetness and light? Recent headlines on research confirming the sweet taste of heavy water and the creation of the lightest isotope of uranium yet may give that impression. But the serious science behind these separate research findings has implications for human health and for the understanding of the process of alpha decay.