Neutron depth profiling technique nurtured at NIST can improve battery technology

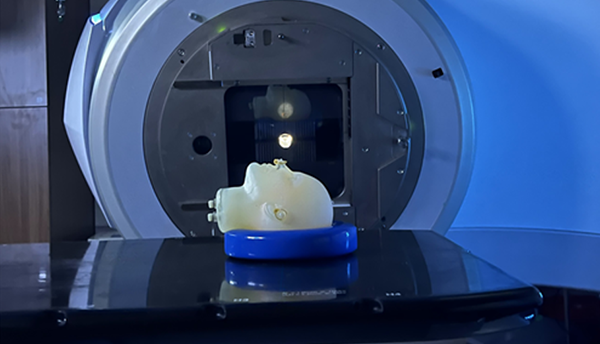
The newest generation of lithium-ion batteries now being developed uses thin-film, solid-state technology and could soon safely power cell phones, electric vehicles, laptops, and other devices. However, like all batteries, solid-state lithium-ion batteries have a drawback: Impedance—electrical resistance—can build up as batteries are discharged and recharged, limiting the flow of electric current.