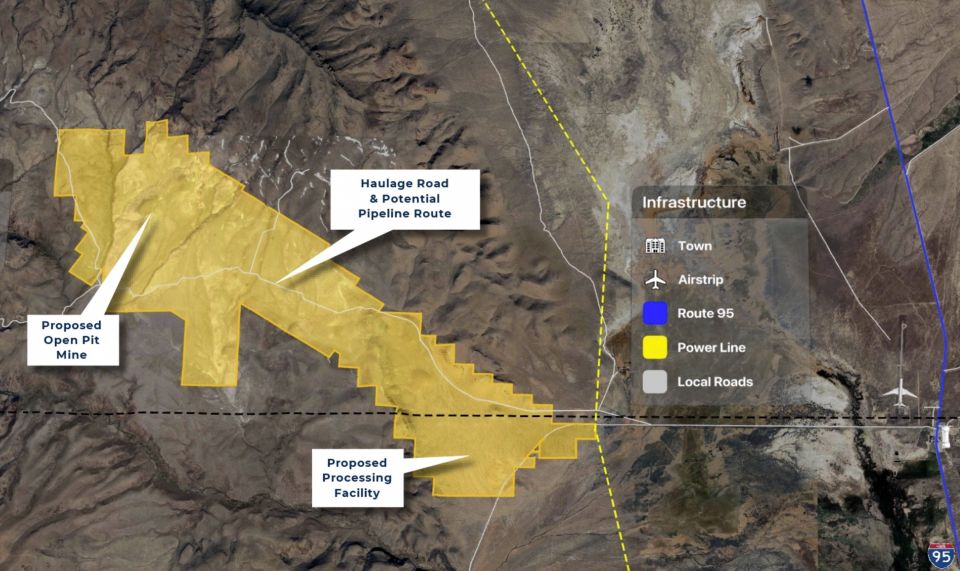
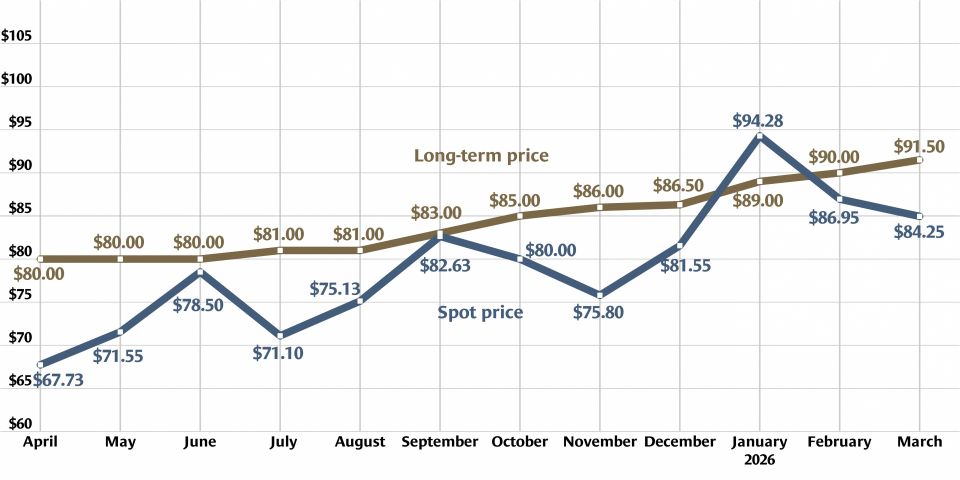
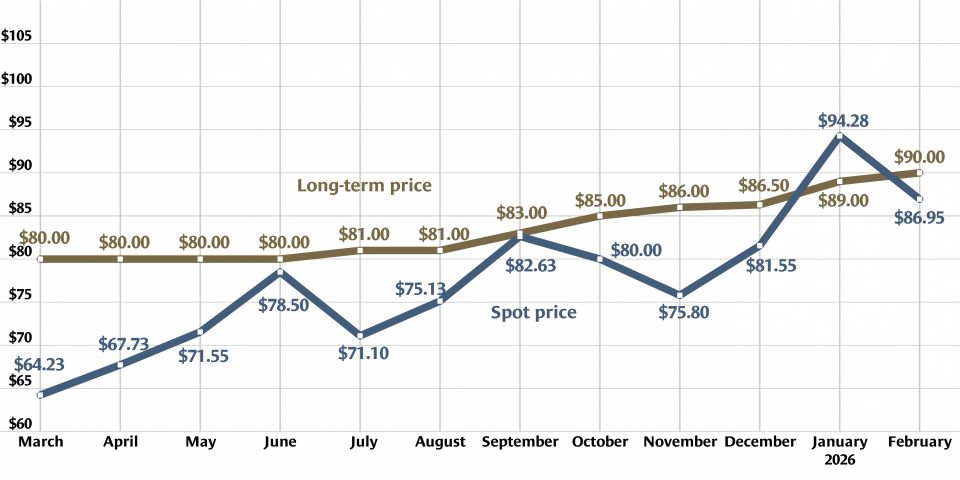
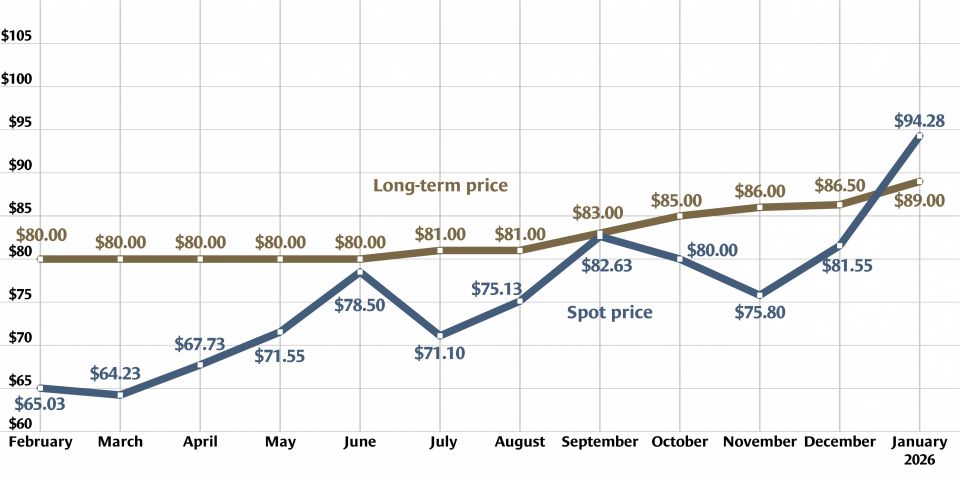
Heavy water, light uranium: One sweet contrast

Is isotope science all sweetness and light? Recent headlines on research confirming the sweet taste of heavy water and the creation of the lightest isotope of uranium yet may give that impression. But the serious science behind these separate research findings has implications for human health and for the understanding of the process of alpha decay.