New adsorbent shows promise in helping remove Cs+ from nuclear wastewater
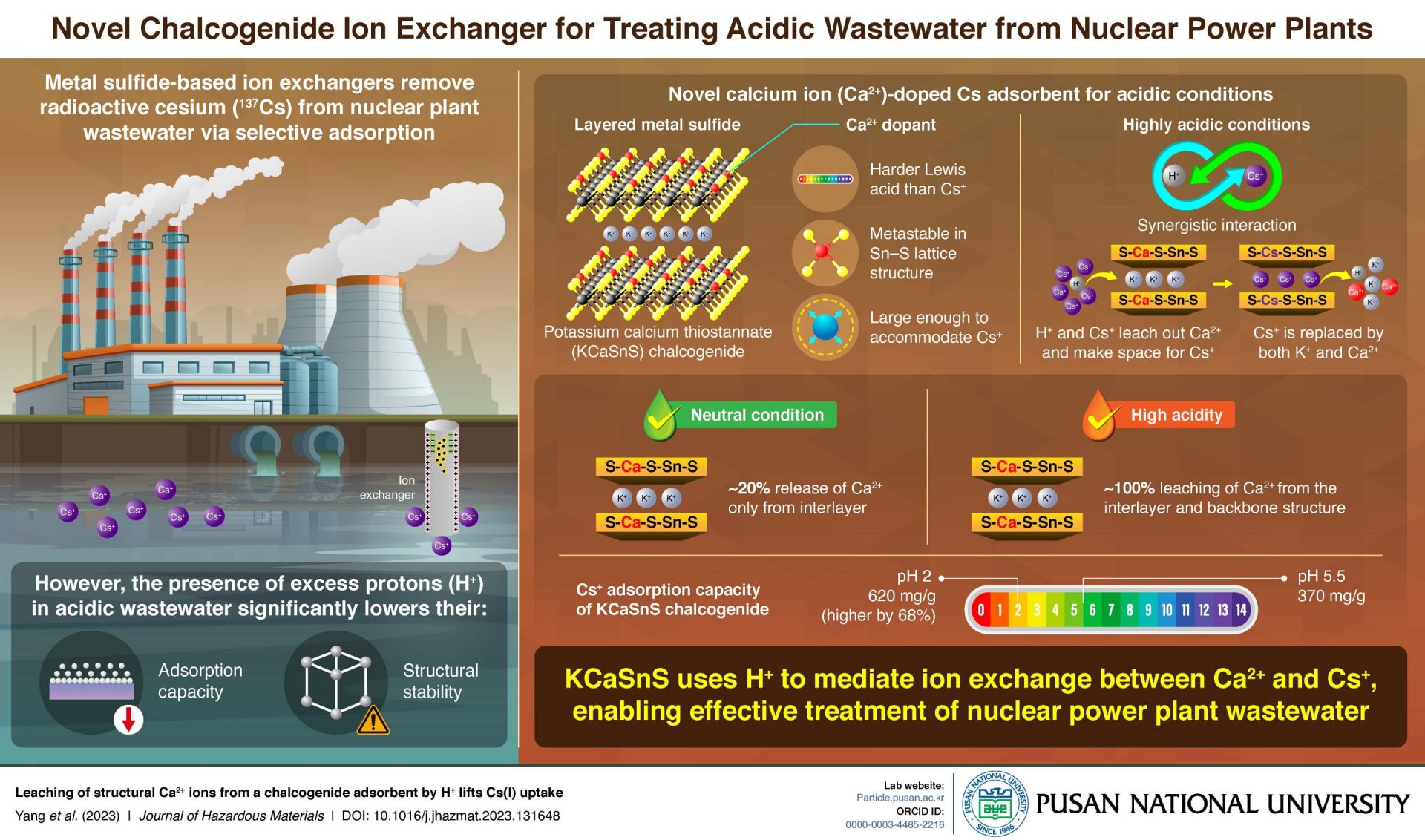
Researchers from the Pusan National University in South Korea have developed a new calcium-doped ion exchanger for the removal of radioactive cesium from acidic nuclear power plant wastewater. The findings have the potential for developing more efficient and effective methods of remediating radioactive contamination.






