Protein shows potential to accelerate cancer therapy research and application
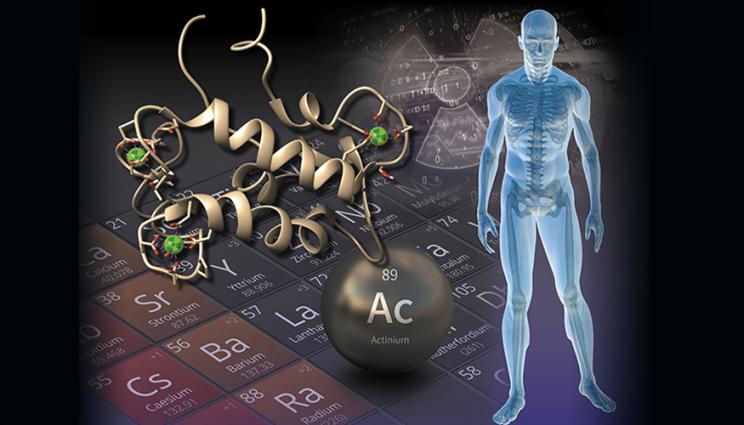
Scientists at Lawrence Livermore National Laboratory and Pennsylvania State University have demonstrated that a natural protein found bonded to rare earth elements can be recovered and used as a tool to purify and effectively manage radioactive metals that show promise for cancer therapy and the detection of illicit nuclear activities.