“The process gave us a lot of development experience, despite all the obstacles we had to overcome,” said Taylor. “We started the project right before the pandemic, and some of us were still trying to get through classes while working on the project.”
The student team worked under Bill Wabbersen, at the time a nuclear engineer at the National Nuclear Security Administration. (Wabbersen retired in September.) “Bill had a goal to create an application that would reach as many users as possible while teaching them about isotopes,” said Taylor. “Together with Bill, we were able to achieve that goal.”
Isotopes are two or more forms of the same element. They contain the same number of protons and so have the same atomic number and position on the periodic table, but they have different numbers of neutrons.
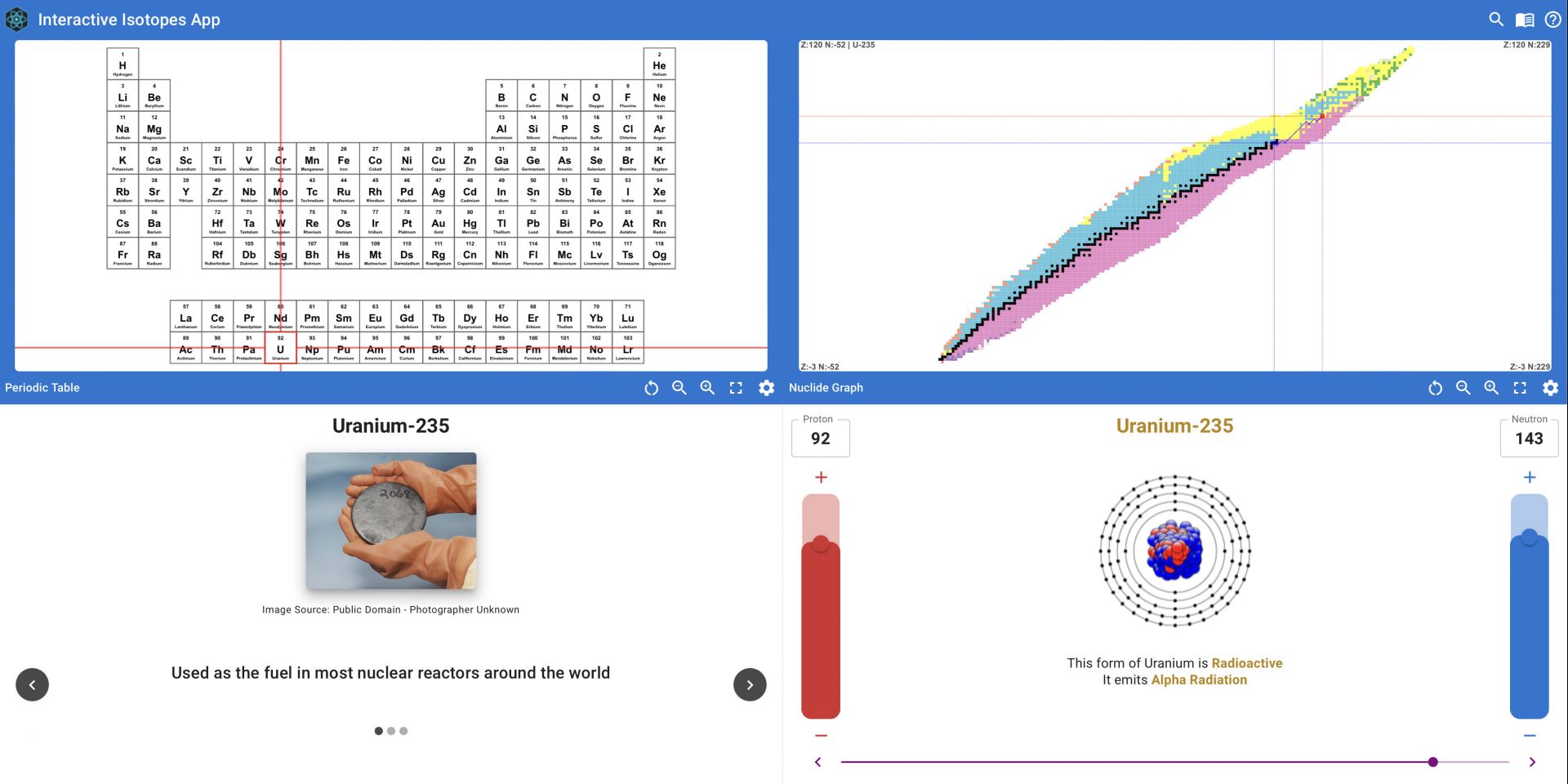
App features: The app features an interactive periodic table. Clicking on an element pulls up specific information in a window that opens, including images (or in some instances, photos of the person who made the discovery) and facts about the uses for their different isotopes. In the absence of known uses, readers are treated to a quip and a bit of history about the element. A nuclide graph that displays all the isotopes of the chemical element is also shown on the main screen for the element selected. In exploring the graph, users can see every known isotope and its position relative to the “line of stability,” where isotopes have the most balanced ratios of protons to neutrons. Radioactive isotopes emit radiation as they rearrange their nucleus in order to become more stable, getting closer to the line of stability over time.
When an element is selected, two vertical bars appear in a window that allow a user to change the number of protons and neutrons. Adding or subtracting neutrons moves only the nuclide graph, since the atomic number remains the same. Adding or subtracting protons also shifts both the nuclide graph and the position on the periodic table. If the isotope selected is radioactive, the decay chain is visible in the nuclide graph. U-235, for example, is the highly radioactive fissile isotope used in nuclear reactors. Over hundreds of millions of years, the U-235 found on earth eventually decays into a stable isotope. Using the dials on the app to enter 92 protons and 143 neutrons will reveal the decay chain for U-235.
In use: The nuclide graph is fully interactive as well. Just like with the periodic table, a user can zoom in and double click to retrieve information or use the sliding bars on the chart at the bottom of the screen.
Zheng, Jones, and Taylor are celebrating the launch of the app as a huge success: “The best way to learn is by doing it, turning the idea into a reality,” said Zheng, now a senior software engineer at Catalyst Consulting Group. “The knowledge gained during the development process is invaluable, and the sense of achievement as we complete the final piece is immeasurable.”
The Interactive Isotopes App started as a project for Android, but when the pandemic slowed development, the team came together and reevaluated the project, ultimately deciding to transition it into an Internet application to reach a wider audience. For functionality, the app can be used either online or offline and is available for desktops and mobile devices. Wabbersen, his colleagues, and volunteers were able to fill in much of the additional isotope data required for the app. The Interactive Isotopes App is available here: https://isotopes.ans.org/.